Master FDA Compliance AuditsInspectionsReadiness
Stay Ready for Every FDA Inspection – Prevent Findings Before They Become 483s
Pharmaceutical, Medical Device and Biologic Visualizations Built For Fast Decisions
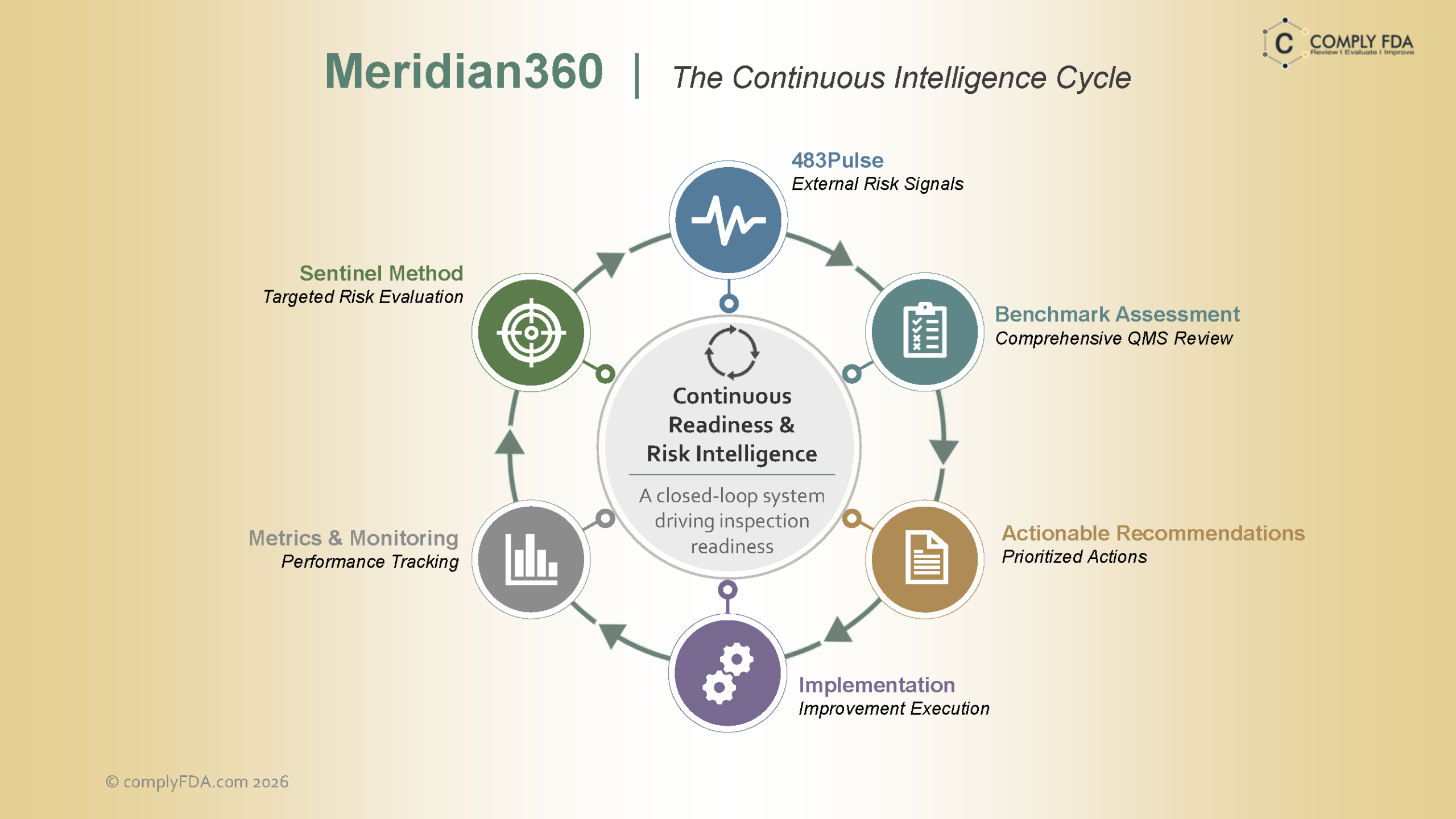
Transforming Regulatory Data Into Clear, Actionable Intelligence
Regulatory data gets complex fast. Our targeted visualizations convert massive datasets into clear, intuitive insights that help executives track compliance performance in real time, anticipate risk, and make confident quality and regulatory decisions without hesitation. Every chart, dashboard, and trend line is engineered for accuracy, speed, and FDA inspection readiness.
Regulatory Visualization Excellence
Turn complex compliance data into clear, interactive visual dashboards that support faster interpretation, stronger oversight, and better long term decision making.
Management Review Intelligence
Give leadership a complete, data backed view of quality performance. Visualize KPIs, audit results, process effectiveness, and regulatory trends so management review meetings lead to decisive action.
Why Life Science Leaders Choose ComplyFDA

what we offer.
Regulatory Intelligence You Can Trust.
Supporting your compliance decisions with precise, data driven insights built for FDA regulated environments.
Our Insider Intelligence products deliver clear, actionable analysis across pharmaceuticals, medical devices, biologics, and combination products. Each solution provides real time visibility into quality trends, inspection risk, and regulatory performance so your team can make confident decisions, reduce compliance gaps, and stay ahead of FDA expectations. Every product is engineered for accuracy, speed, and long term compliance success.
Your path to stronger regulatory confidence
Create Inspection Readiness, Strengthen Compliance Decisions, and Improve Quality Outcomes.
Life science organizations rely on fast, accurate intelligence to stay ahead of FDA expectations. Our Insider Intelligence platform delivers clear visibility into compliance trends, risk signals, and quality system performance so your team can act with precision and maintain continuous readiness.
Get targeted regulatory insights built for your quality and compliance goals.
Schedule a Compliance Consultation

Always stay inspection ready with clear visibility into your compliance data.
Review Your Quality System With Confidence.
Our platform helps teams review compliance data quickly and accurately. Identify signals across audits, CAPA activity, and production trends so you always know where you stand with FDA expectations.
Stronger regulatory outcomes start with precise evaluation.
Evaluate Risks, Trends, and Performance With Clarity.
Use targeted insights to evaluate quality metrics, recurring deviations, and risk profiles across products and processes. Get the clarity needed to support confident decisions in a regulated environment.


A continuous improvement mindset that drives better compliance.
Improve Processes With Actionable Intelligence.
Optimize your quality system by turning data into real improvements. Strengthen procedures, reduce compliance gaps, and support long term inspection readiness with insights that guide your next step.
Client Reviews.
Trusted by Life Science Teams Committed to Stronger Compliance.
Rating 4.9 / 5.0 (verified across multiple platforms)