Foundational Compliance Intelligence
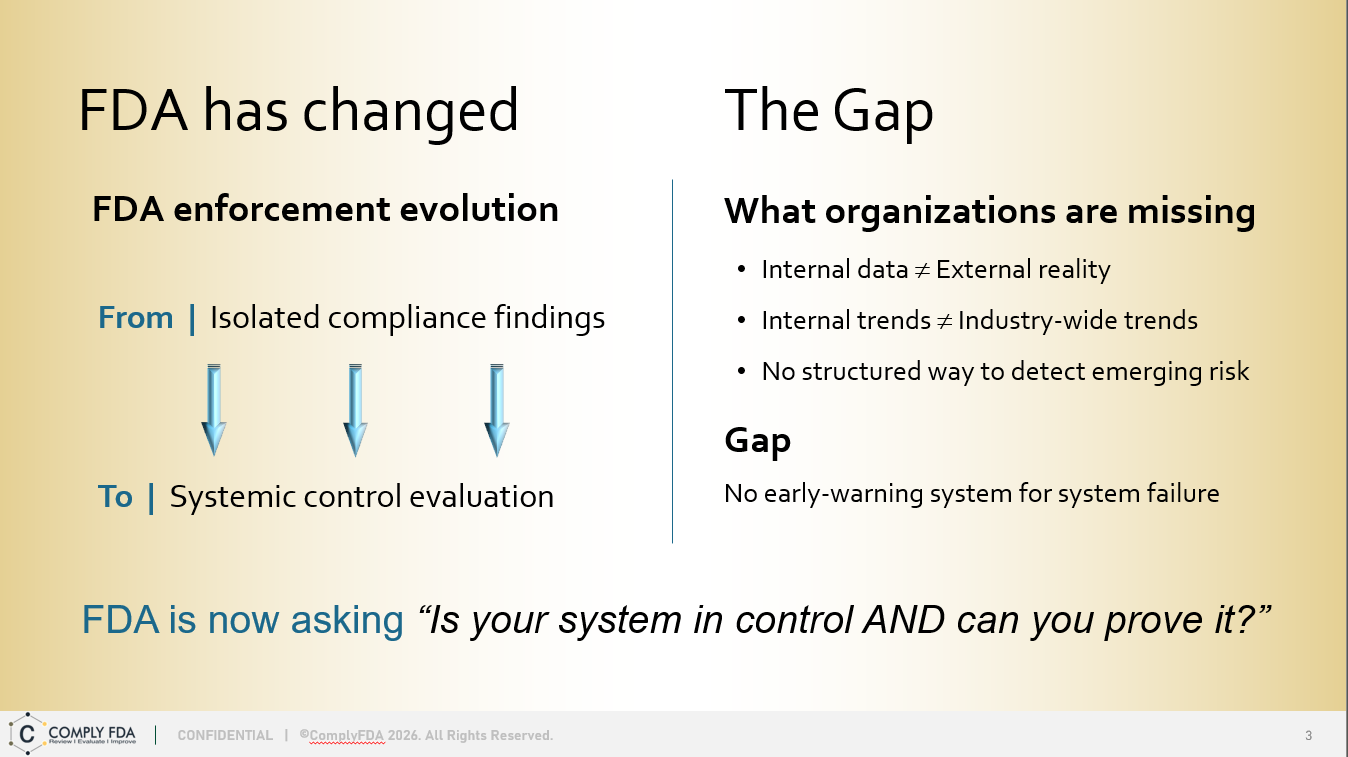
SHIFTING THE INSPECTION READINESS PARADIGM
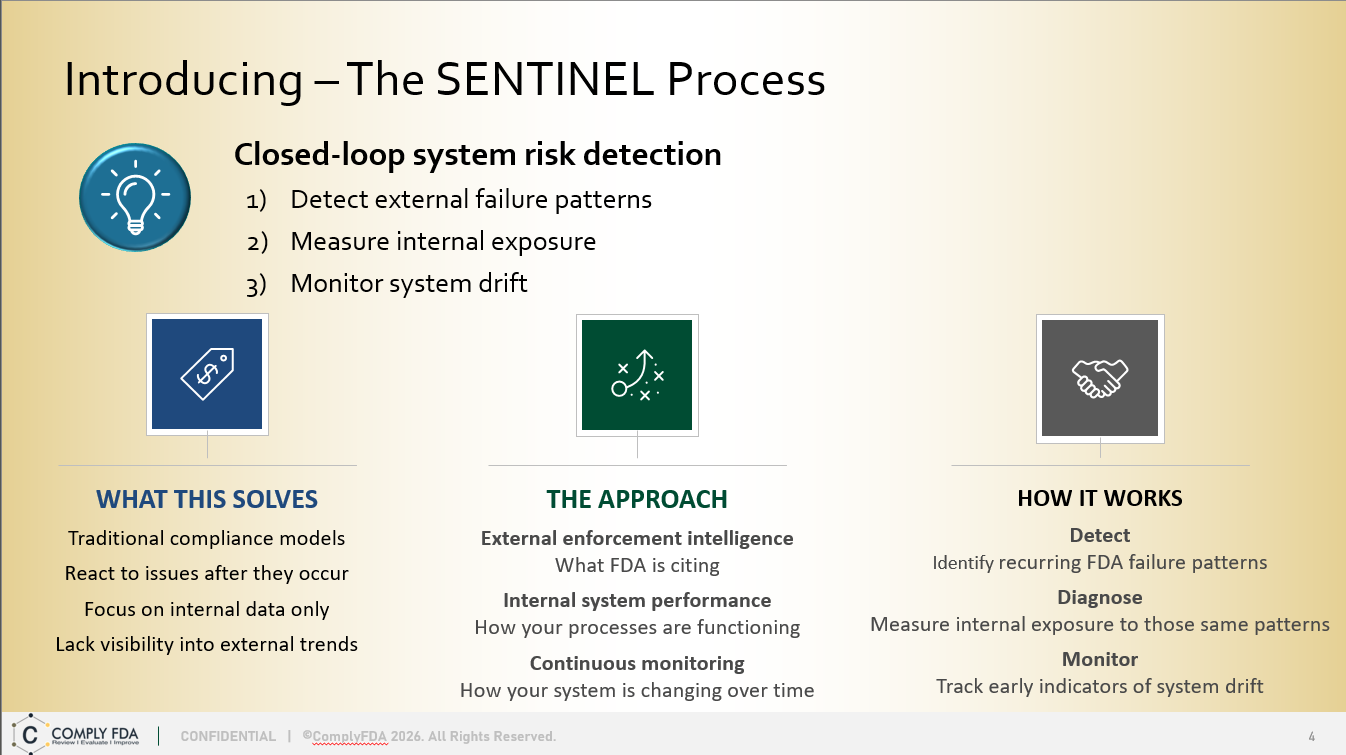
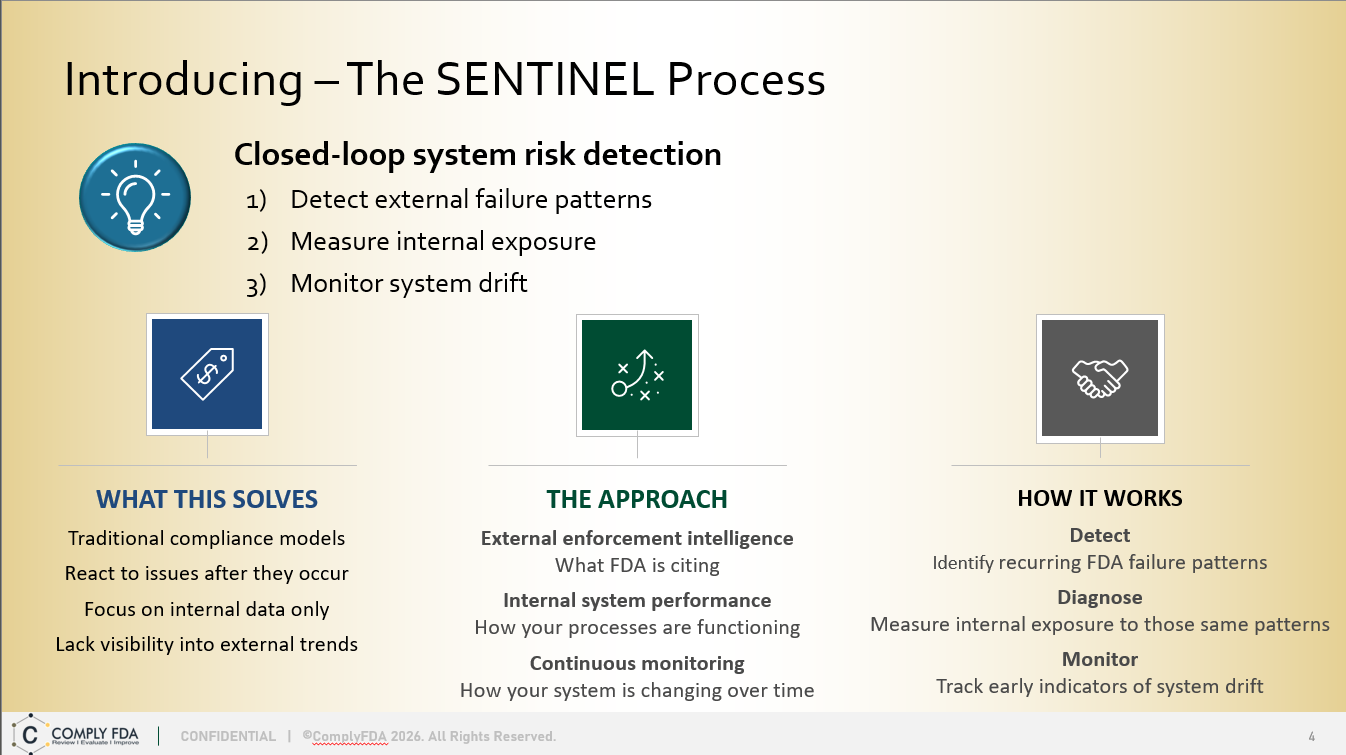
The SENTINEL Method is a strategic quarterly quality system assessment service — See What’s Coming. Act Before It Arrives. It combines FDA 483 trend intelligence with a best practice framework to deliver predictive, efficient, and actionable compliance risk reduction for budget-conscious organizations. Assessments performed for one or more quarters.
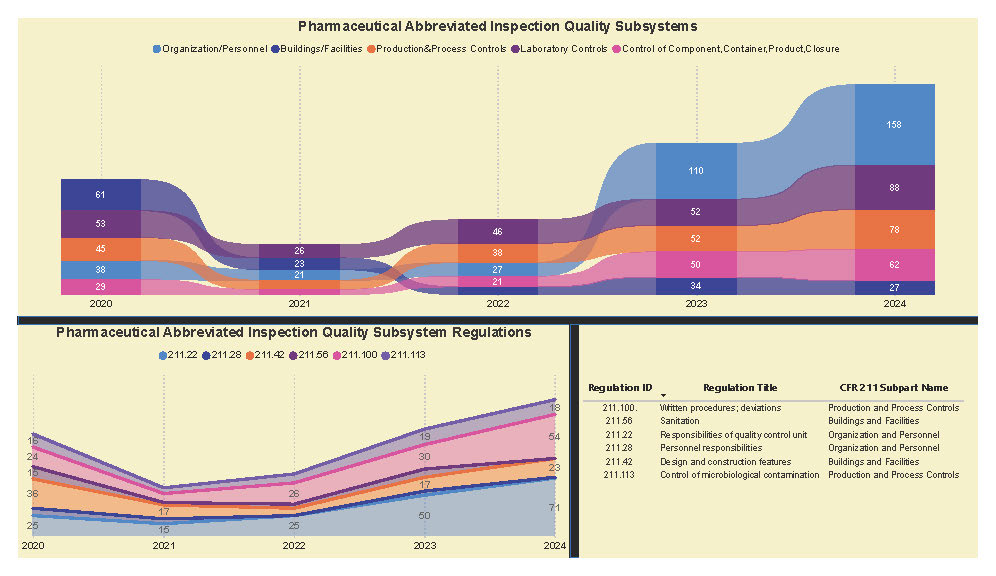
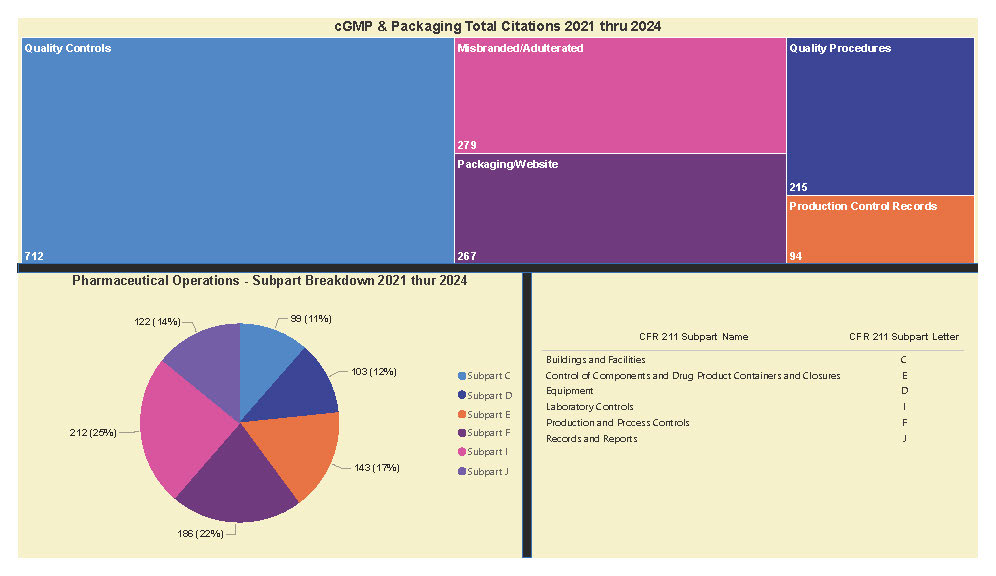
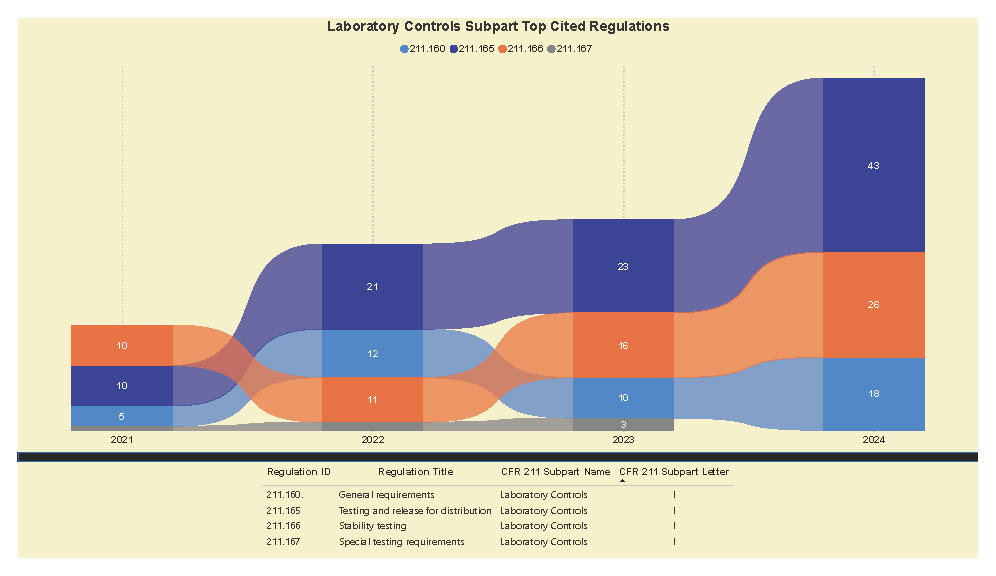
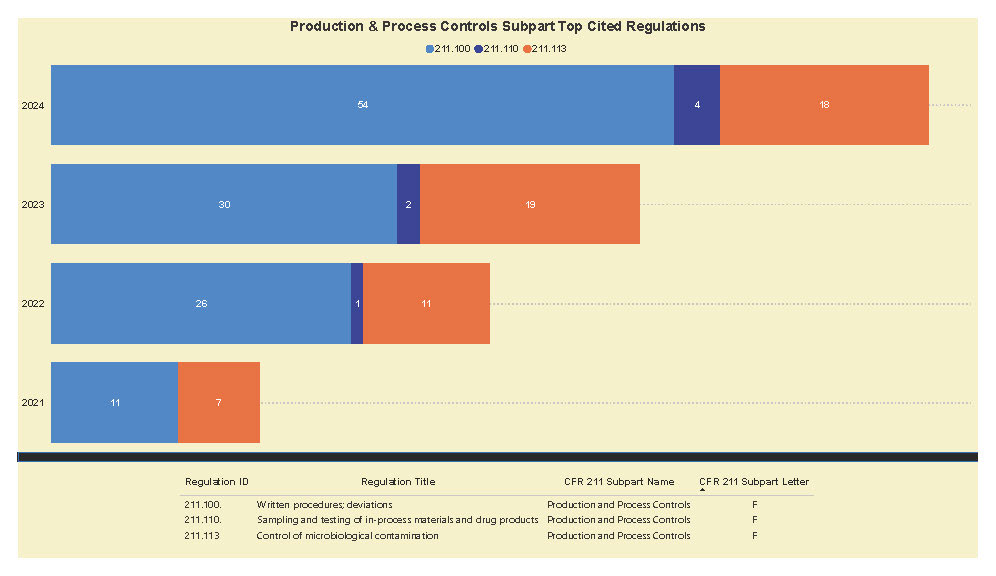
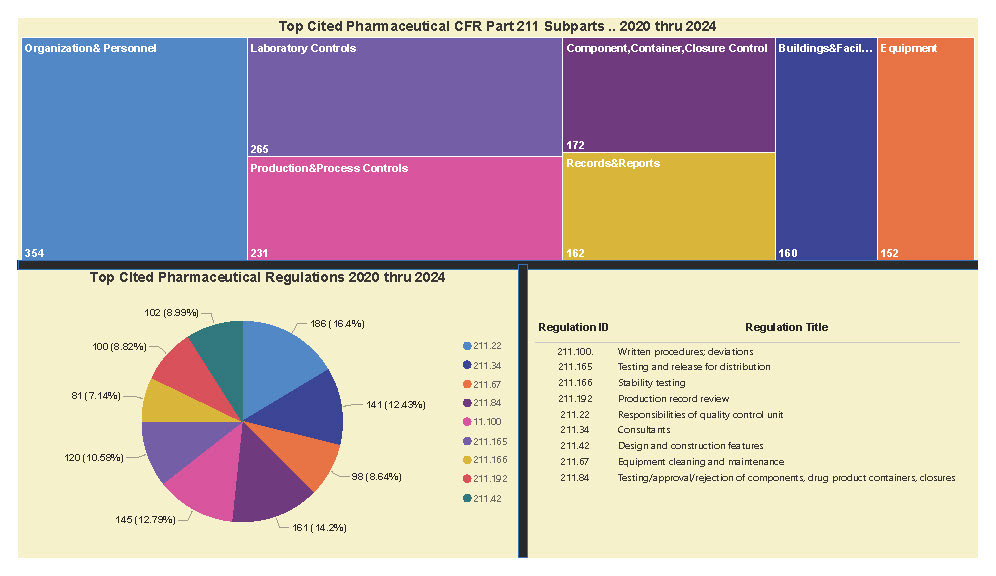
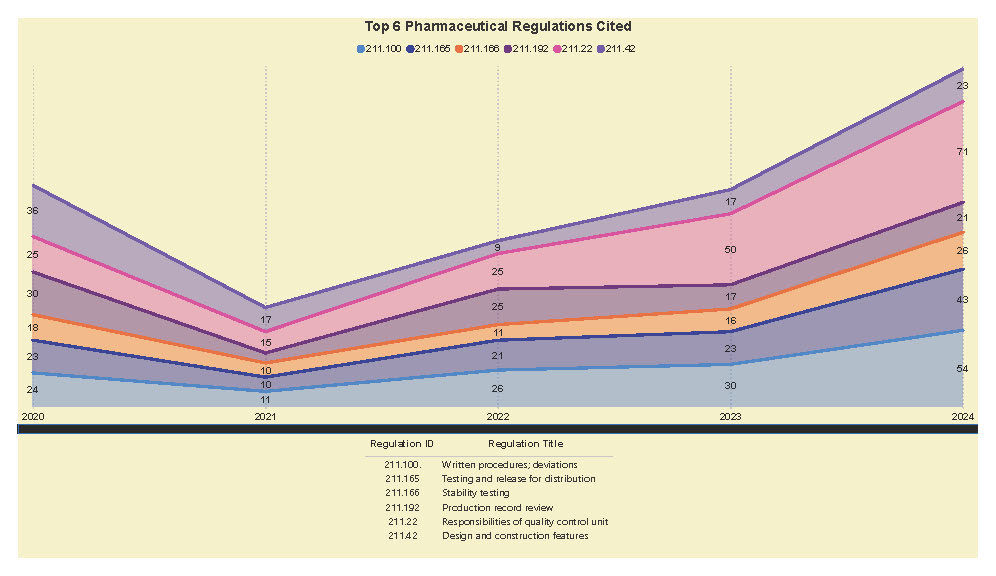
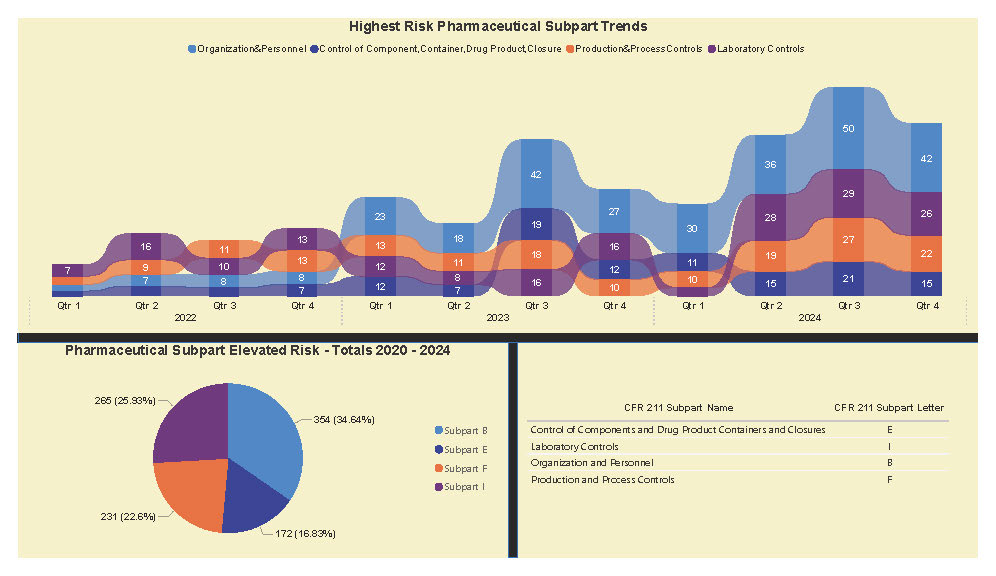
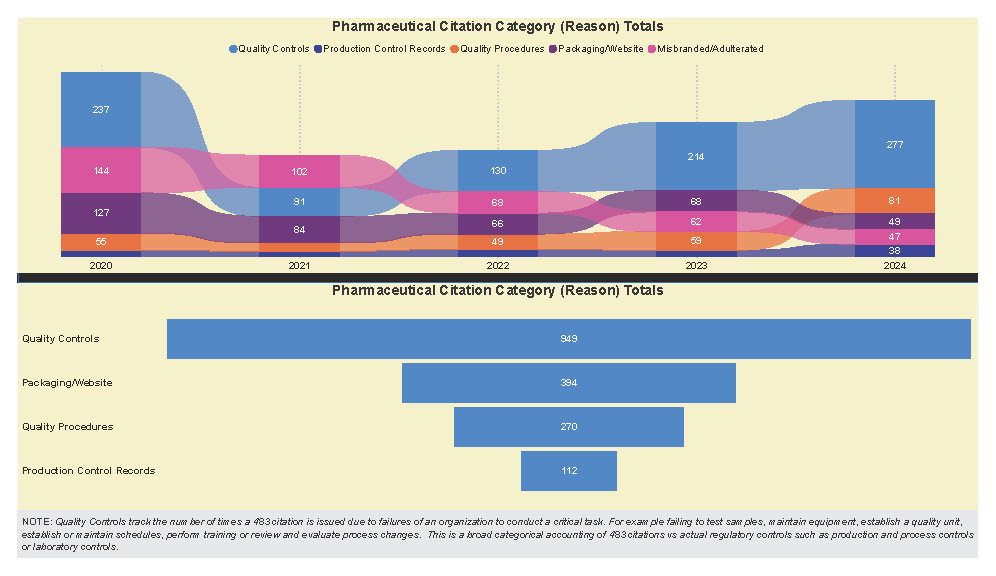
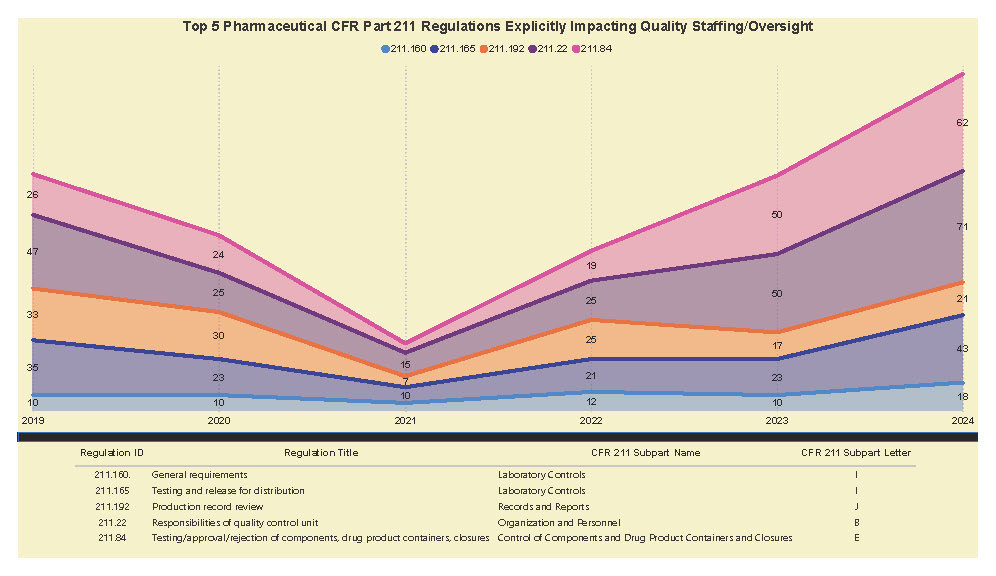
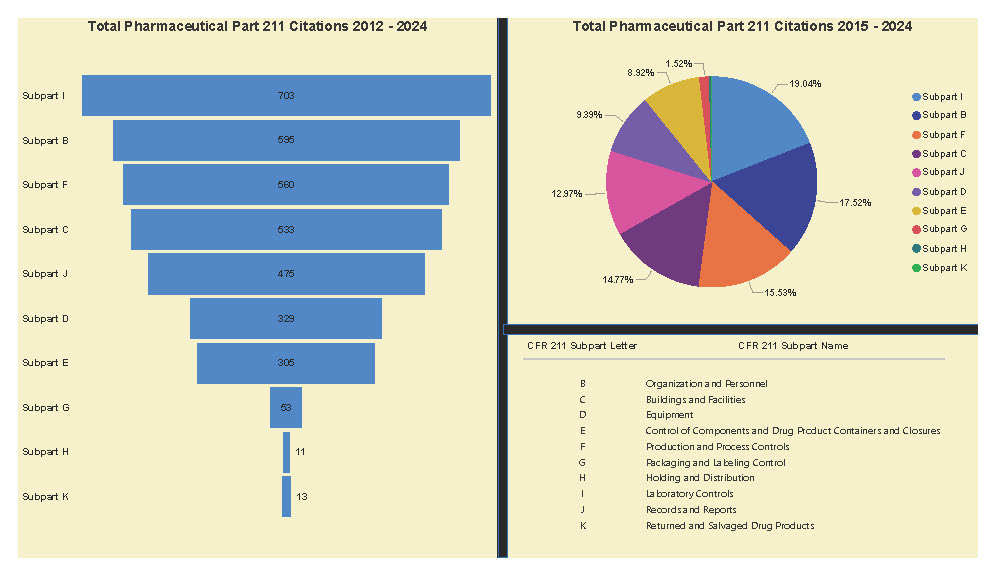
A Subpart Analysis is a comprehensive report of all FDA 483 activity from 2017 through present for both pharmaceuticals (PharmaBenchmark) and devices (DeviceBenchmark). All Part 211 and Part 820 regulations are grouped by subpart with active links to current eCFR guidance. Each subpart is organized by regulation showing current 483 links from 2017 leveraging our proprietary database of FDA enforcement activity.
Current subscribers frequently cite the extensive research behind this resource and the concise way the information is presented. Organizations use it to benchmark performance, evaluate supplier opportunities, conduct risk analysis, and support both management reviews and strategic planning. Subscribers report discovering new applications for the data as they integrate it into their decision-making processes.
See the redacted example below.
| PART 211 CURRENT GOOD MANUFACTURING PRACTICE FOR FINISHED PHARMACEUTICALS |
| Subpart B–Organization and Personnel |
| § 211.22 – Responsibilities of quality control unit. |
| § 211.25 – Personnel qualifications |
| § 211.28 – Personnel responsibilities. |
| § 211.34 – Consultants |