GxP Assessments
FDA Compliance Best Practices for Quality Systems, Design Controls, and Inspection Readiness
Practical FDA Compliance Best Practices Built for Real Inspections
If you are responsible for quality, regulatory affairs, or compliance, the question is rarely whether you have procedures. The real question is whether your systems and quality processes are connected as a “system-level” capability including risk management. Robust quality management system processes are the foundation for addressing FDA’s core issue “Is your system in control AND can you prove it”? This page outlines FDA compliance best practices that are built from actual inspection behavior, common Form 483 observations, and enforcement trends, not theory.
These quality process best practices are designed for pharmaceutical, medical device, biologics, and combination product organizations that want fewer surprises, shorter inspections, and defensible compliance decisions that stand up under scrutiny from the FDA and notified bodies.
ComplyFDA’s approach is grounded in real GxP assessments, regulator-aligned benchmarking, and inspection-tested methodologies used by companies that operate in high-risk, high-visibility regulatory environments.
Quality Management System Best Practices the FDA Expects to See
A compliant Quality Management System is defined not by the number of SOPs, but whether it works consistently, is followed in practice, and can be demonstrated to be in control during an inspection. Effective FDA quality systems require clear ownership, documented decision-making, and traceability across procedures, records, and outcomes. Alignment with validated best practices signals robust process performance investigators now actively assess. Inspectors frequently cite firms where quality systems exist on paper but fail in execution. FDA-regulated organizations outperforming peers build quality systems that demonstrate control before anyone asks.
Key quality system best practices that reduce inspection risk include:
- Establish clear process ownership which signals systemic strength
- End-to-End traceability with formal change management; validated and controlled changes
- Robust CAPA systems that provide credible root cause and demonstrate effectiveness
- Rigorous internal audits and regular management reviews using measurable quality metrics
Organizations that struggle during inspections often discover gaps between what is written and what is performed. Closing that gap is one of the fastest ways to reduce FDA inspection risk.
Documentation and Data Integrity Best Practices
Documentation failure signals a regulated organization cannot account for what it does, when it did it, or who authorized it — which is why documentation findings consistently top FDA inspection outcomes.
The risk is most acute in electronic environments. Audit trails, user access controls, and data retention policies are now standard inspection targets. Organizations that treat these as IT responsibilities rather than quality system requirements remain consistently vulnerable.
High-performing organizations are defined by a leadership-driven expectation that every record is accurate, complete, and traceable at the moment of creation. Aligning documentation standards with current industry best practices doesn’t just reduce inspection risk — it builds the institutional credibility that accelerates approvals, strengthens regulatory relationships, and protects revenue. Embedded into daily operations, that standard is what FDA investigators recognize as a system genuinely in control, and what delivers a measurable competitive advantage.


Design Control and Risk Management Best Practices
Design controls remain one of the most scrutinized inspection domains, with findings increasingly harder to defend. FDA investigators are not simply checking whether design documentation exists. They are tracing decision integrity from user need to validated output, exposing every gap where risk management was treated as a document versus a discipline.
The organizations most exposed are those where risk files were built to satisfy a submission and never revisited. When designs change, complaints surface, or post-market data signals emerging risk, an unchanged risk file is not a defensible position. It demonstrates risk management is disconnected from how the product actually performs.
Organizations that align design controls and risk management with current best practices; ISO 14971 integration, traceable design history, and post-market feedback loops that actively inform design decisions, operate from a position of demonstrable control. That alignment accelerates development cycles, strengthens submission credibility, and brings safer products to market faster. In a regulatory environment where scrutiny of design history is intensifying, that is not a compliance investment. It is a commercial one.
Inspection Readiness Best Practices That Reduce Form 483 Findings
The organizations most vulnerable during an FDA inspection are rarely those with the weakest quality systems. They are those whose systems were never tested under inspection conditions. Readiness is a capability, and like every capability, it degrades without deliberate practice.
Successful leaders treat inspection readiness as continuous. Structured internal audits aligned to current FDA enforcement trends and staff who can answer investigator questions without escalation replace reactive defense with demonstrated control. The return is measurable: vulnerabilities identified and corrected before regulators arrive, shorter inspection timelines, and sustained regulatory standing that protects approvals, market access, and commercial continuity.
In today’s enforcement environment, inspection readiness is not a quality function responsibility. It is a leadership performance standard.
ComplyFDA routinely helps organizations identify inspection vulnerabilities before the FDA does. Corrective action happens on your timeline, not under regulatory pressure.

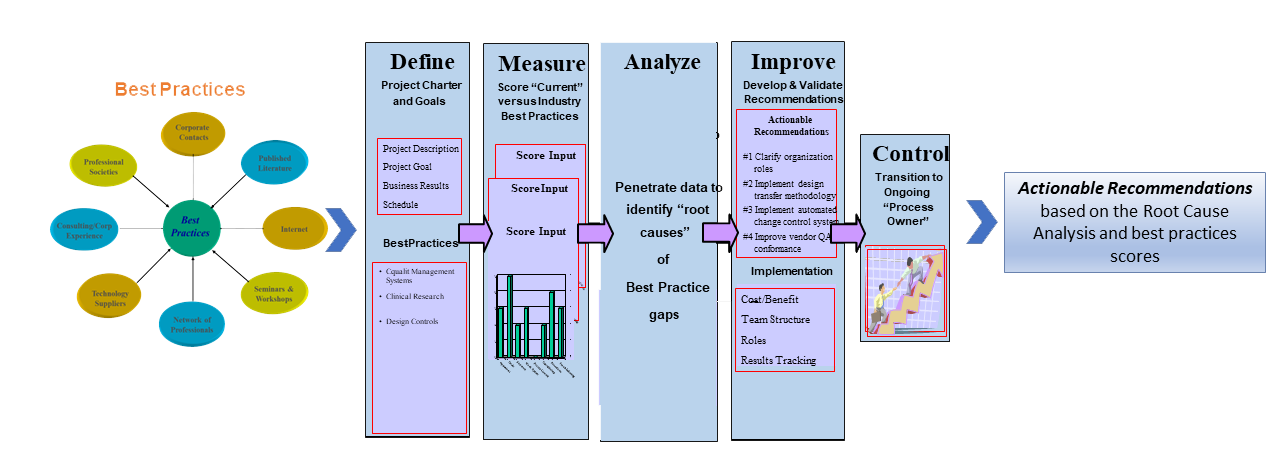
How ComplyFDA Defines and Applies Best Practices
ComplyFDA’s best practices are not generic templates. They are derived from structured GxP assessments, regulatory benchmarking, and ongoing analysis of inspection outcomes across the life sciences industry.
Our teams evaluate quality systems, clinical processes, and design controls against FDA expectations, industry standards, and real inspection behavior. Actionable recommendations are prioritized based on risk, regulatory impact, and operational feasibility.
This approach ensures that improvements are defensible, practical, and aligned with how inspectors actually evaluate compliance, including those from the U.S. Food and Drug Administration.
Integrating validated best practices demonstrates quality process control and ongoing quality process performance monitoring.
FDA Compliance Best Practices FAQs